)
Are you reacting too late to water quality changes?
Are you reacting too late to water quality changes?
Application insight for mining and metal processing operations
In many industrial water treatment systems, process control relies on a combination of online measurements and laboratory analysis.
While this approach provides some visibility to water quality, it often does not give enough information at the moment when decisions need to be made.
How water quality is typically monitored
Most operations continuously monitor parameters such as pH, conductivity and sometimes redox potential. These measurements are useful for detecting that something is changing in the system. However, they do not explain what is changing or whether the change requires action.
At the same time, dissolved metals — which are often critical for treatment performance and compliance — are typically measured through laboratory analysis. In many cases, composite samples are taken over the day and analysed once per day, with results available the next day.
This means that the actual chemical state of the water is often confirmed only after the process has already evolved.
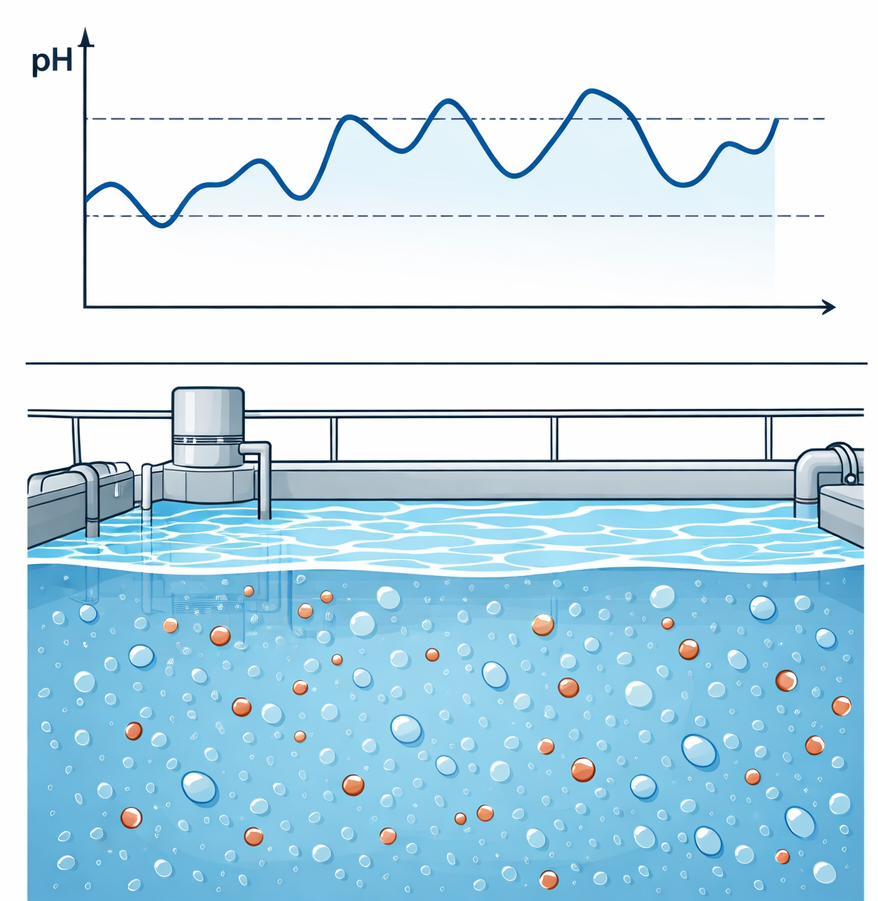 Fig: Online measurements show that something is changing — but not what is changing.
Fig: Online measurements show that something is changing — but not what is changing.
What this means in practice
When treatment performance depends on dissolved metals, operators are often required to make decisions without having full visibility into the current situation.
If online signals indicate a change, the response is typically based on experience and safety margins rather than confirmed data. By the time laboratory results are available, the situation may already have changed.
This creates a gap between process control and actual water chemistry.
Why this matters
When process decisions are made without clear visibility into dissolved metals, the consequences are often practical and immediate:
increased risk of non-compliance
over-treatment of water to maintain safety margins, leading to unnecessary chemical consumption
unnecessary process adjustments
operational uncertainty in daily decision-making
Example of potential impact of over-treatment of wastewater:

A different approach
Instead of relying only on delayed laboratory results, operations are starting to look at faster access to dissolved metal information directly at the site.
With earlier insight into water chemistry, it becomes possible to better understand whether observed changes require action and how the treatment process should be adjusted.
The key is not only detecting that something is changing — but understanding what is changing and when.
Where this is being explored
Rapid dissolved-metal analysis is being evaluated in operations where:
compliance requirements are strict
treatment performance depends on metal concentrations
delayed information creates uncertainty in process control
In these cases, the goal is to reduce the gap between measurement and decision-making.
Let’s discuss your case
Every water treatment system is different.
If you are interested in improving visibility into your water chemistry or reducing uncertainty in process control, it would be interesting to exchange experiences.
You can contact us directly at: tuomo@3awater.com